Mri Medical Device Classification . Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. Food and drug administration's current recommendations for mr safety terminology. This editorial presents the u.s. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. I, iia, iib and iii. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes:
from www.slideshare.net
Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: This editorial presents the u.s. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. I, iia, iib and iii. Food and drug administration's current recommendations for mr safety terminology.
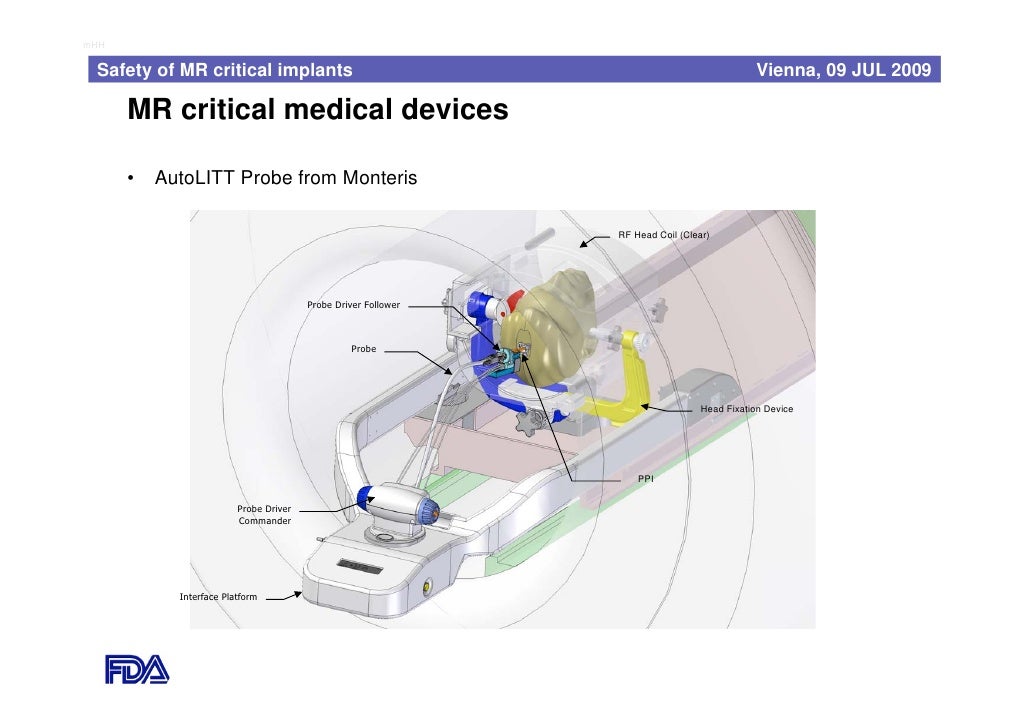
Safety of MRI Critical Medical Devices
Mri Medical Device Classification A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: This editorial presents the u.s. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. I, iia, iib and iii. Food and drug administration's current recommendations for mr safety terminology.
From wtop.com
New MRI helps patients with implanted devices, claustrophobia or Mri Medical Device Classification This editorial presents the u.s. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. I, iia, iib and iii. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: This document provides guidance to assist product owners in risk classification of in. Mri Medical Device Classification.
From www.biomedicalviews.com
Types of MRI Scanners Mri Medical Device Classification Food and drug administration's current recommendations for mr safety terminology. I, iia, iib and iii. This editorial presents the u.s. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23.. Mri Medical Device Classification.
From www.tuvsud.com
Infographic MRI Safety and Compatibility Testing TÜV SÜD Mri Medical Device Classification This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. This editorial presents the u.s. I, iia, iib and iii. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: A framework for device classification for clinical research protocols using mri in the. Mri Medical Device Classification.
From www.ejradiology.com
MRI with cardiac pacing devices Safety in clinical practice Mri Medical Device Classification Food and drug administration's current recommendations for mr safety terminology. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. This editorial presents the u.s. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. This document provides guidance to. Mri Medical Device Classification.
From www.verywellhealth.com
How an MRI Machine Works for Orthopedics Mri Medical Device Classification Food and drug administration's current recommendations for mr safety terminology. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: Applications for mr conditionality shall be evaluated for impact to all general safety. Mri Medical Device Classification.
From www.nibib.nih.gov
Resonance Imaging (MRI) National Institute of Biomedical Mri Medical Device Classification This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. A framework for. Mri Medical Device Classification.
From meddev-info.blogspot.com
Medical Device Regulation Basics US FDA Medical Device Classification Mri Medical Device Classification I, iia, iib and iii. This editorial presents the u.s. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: Food and drug administration's current recommendations for mr safety terminology. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. This document provides. Mri Medical Device Classification.
From www.ejradiology.com
MRI with cardiac pacing devices Safety in clinical practice Mri Medical Device Classification Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: This editorial presents the u.s. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. Food and drug administration's current recommendations for mr safety terminology. A framework for device classification for clinical research. Mri Medical Device Classification.
From www.technicalprospects.com
Siemens MRI Training Mri Medical Device Classification This editorial presents the u.s. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. Food and drug administration's current recommendations for mr safety terminology. I, iia, iib and iii. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: Applications for mr. Mri Medical Device Classification.
From bookmerilab.com
MRI Defecography Price, Purpose, Procedure & Results [2024] Mri Medical Device Classification Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. A framework for. Mri Medical Device Classification.
From www.slideshare.net
Safety of MRI Critical Medical Devices Mri Medical Device Classification This editorial presents the u.s. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Food and drug administration's current recommendations for mr safety terminology. This document provides guidance to assist product owners. Mri Medical Device Classification.
From www.nuchoiceimaging.com
Difference Between CT & MRI — NuChoice Imaging Mri Medical Device Classification Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. I, iia, iib and iii. This editorial presents the u.s. Food and drug administration's current recommendations for mr safety terminology. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23.. Mri Medical Device Classification.
From www.neuromodulation.abbott
MRI Scans with Your Neurostimulation Device Abbott Mri Medical Device Classification I, iia, iib and iii. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. This editorial presents the u.s. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Food and drug administration's current recommendations for mr safety terminology.. Mri Medical Device Classification.
From www.slideserve.com
PPT Resonance Imaging (MRI) PowerPoint Presentation ID1269956 Mri Medical Device Classification I, iia, iib and iii. This editorial presents the u.s. Food and drug administration's current recommendations for mr safety terminology. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the.. Mri Medical Device Classification.
From debuggercafe.com
Brain MRI Classification using PyTorch Mri Medical Device Classification This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. This editorial presents the u.s. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. I, iia, iib and iii. A framework for device classification for clinical research protocols using. Mri Medical Device Classification.
From fdocuments.in
MRI Patient Guidelines Axonics · MRI Patient Guidelines For patients Mri Medical Device Classification Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. This document provides. Mri Medical Device Classification.
From www.mdpi.com
Diagnostics Free FullText An Enhanced Machine Learning Approach Mri Medical Device Classification A framework for device classification for clinical research protocols using mri in the in vivo nmr center revised march 23. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. Under the mdr, which went into effect on 26 may 2021, devices are divided into four classes: I, iia, iib. Mri Medical Device Classification.
From theaisummer.com
Deep learning in MRI beyond segmentation Medical image reconstruction Mri Medical Device Classification I, iia, iib and iii. This document provides guidance to assist product owners in risk classification of in vitro diagnostic (ivd) medical devices using the. Applications for mr conditionality shall be evaluated for impact to all general safety and performance requirements (gsprs) of the eu. Under the mdr, which went into effect on 26 may 2021, devices are divided into. Mri Medical Device Classification.